Localization of D-AA-related enzymes
In situ hybridization (ISH)
A given sequence of nucleic acids (RNA/DNA) can be localized in tissue using the ISH approach. This is generally accomplished by applying a probe strand of nucleic acids which is complementary to the target strand and labeled in some way to enable observations of the target localization in the tissue of interest. In this way, RNA or DNA corresponding to proteins connected to D-AAs, such as those related to known racemases or D-form amino acid oxidases, can be visualized.
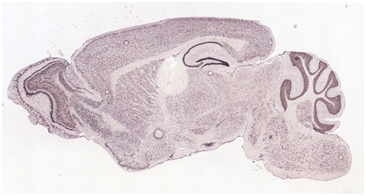
Fig. 1. ISH demonstrating the distribution of serine racemase expression in an adult mouse brain slice. Image acquired from the Allen Brain Atlas.
http://mouse.brain-map.org/experiment/show?id=74357621 (accessed Dec 29, 2016).
Immunohistochemistry (IHC)
For studying the localization of proteins associated with the D-AAs (such as a racemase responsible for the synthesis of a given D-AA), fluorescence imaging following sample treatment with antibodies is often employed. Whereas the ISH approach targets strands of nucleic acids, IHC is generally used to measure proteins that have been translated – avoiding staining of cells/regions which contain the corresponding DNA/RNA but not the translated proteins. Figure 2 shows a whole-mount immunohistochemical stain of the D-aspartate racemase (DAR1) in Aplysia californica.
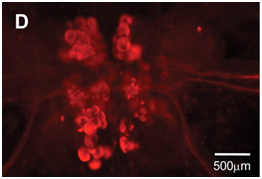
Localization of enzyme activity
ISH can localize specific strands of nucleic acids and IHC can be used to image translated proteins, but one can also treat tissue with carefully selected reagents for imaging on the basis of enzyme activity. This was eloquently demonstrated by Sasabe and coworkers with D-amino acid oxidase (DAAO) in mouse tissue (Fig. 3).
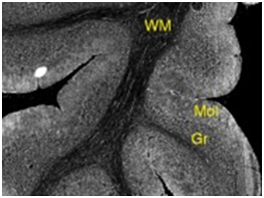
Image adapted from (Sasabe et al., Front Synaptic Neurosci., 2014, 6:14).
References
Immunohistochemistry (IHC):
Wang L., Ota N., Romanova E.V., Sweedler J.V. A novel pyridoxal 5’-phosphate-dependent amino acid racemase in the Aplysia californica central nervous system. J. Biol. Chem., 2011, 286(15), 13765–13774
Localization of enzyme activity:
Sasabe J., Suzuki M., Imanishi N., Aiso S. Activity of D-amino acid oxidase is widespread in the human central nervous system. Front. Synaptic Neurosci., 2014, 6(14)
Author
Amil Patel and Jonathan Sweedler, University of Urbana – Champaign
